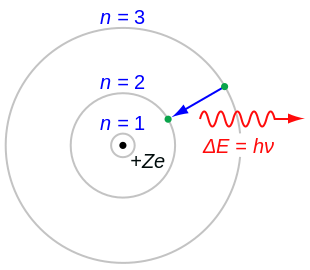
<aside> 🦓 The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell model(价电子层理论). As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory.
</aside>

The Bohr model gives almost exact results only for a system where two charged points orbit each other at speeds much less than that of light.
To overcome the problems of Rutherford's atom, in 1913 Niels Bohr put forth three postulates that sum up most of his model:
<aside> ✅ - 【定态】stationary orbits
</aside>
<aside> ✅ Several enhancements to the Bohr model were proposed, most notably the Sommerfeld or Bohr–Sommerfeld models, which suggested that electrons travel in elliptical orbits around a nucleus instead of the Bohr model's circular orbits.[1] This model supplemented the quantized angular momentum condition of the Bohr model with an additional radial quantization condition, the Wilson–Sommerfeld quantization condition[42][43]
</aside>
$$ \int_{0}^{T}p_{r}dq_{r}=nh, $$